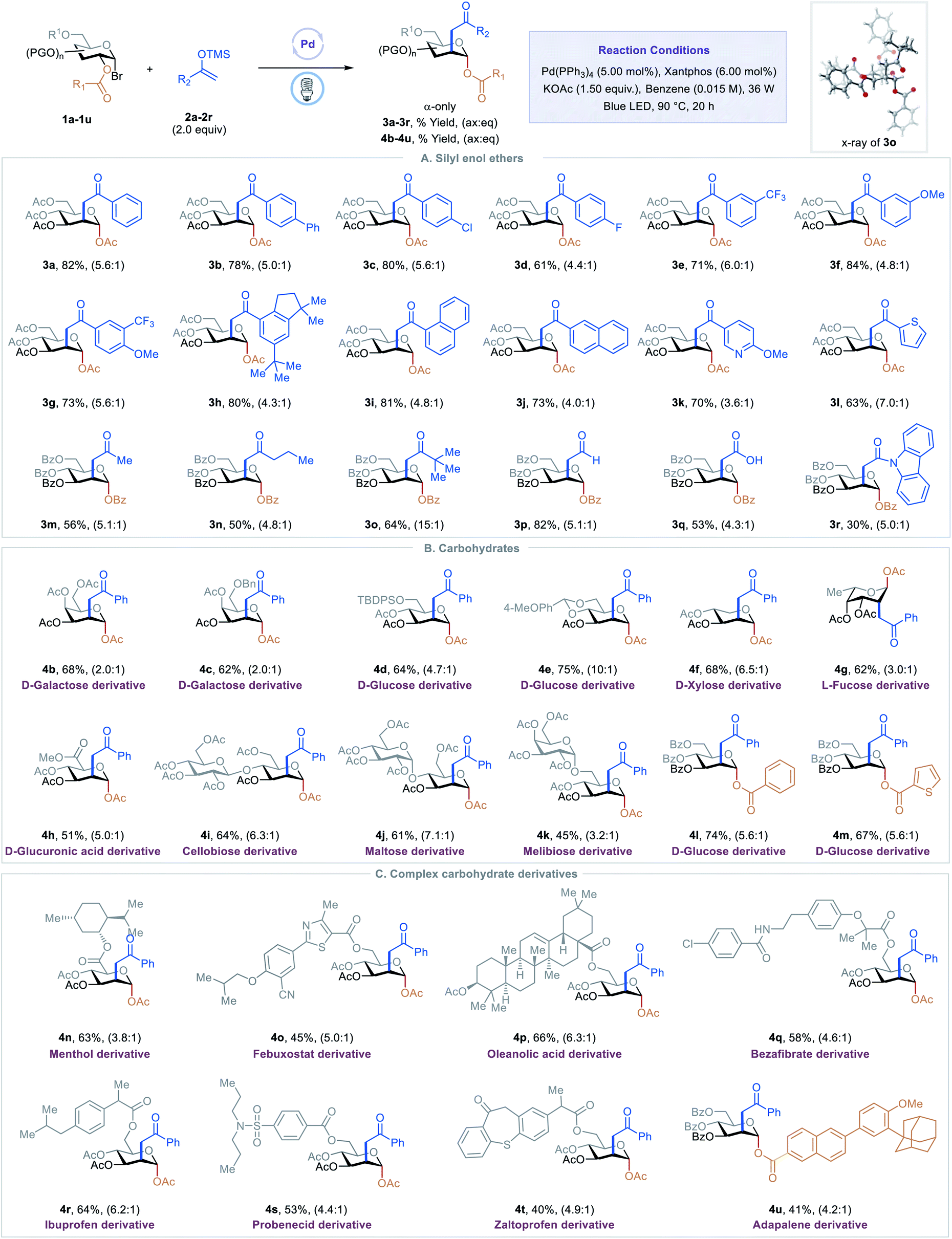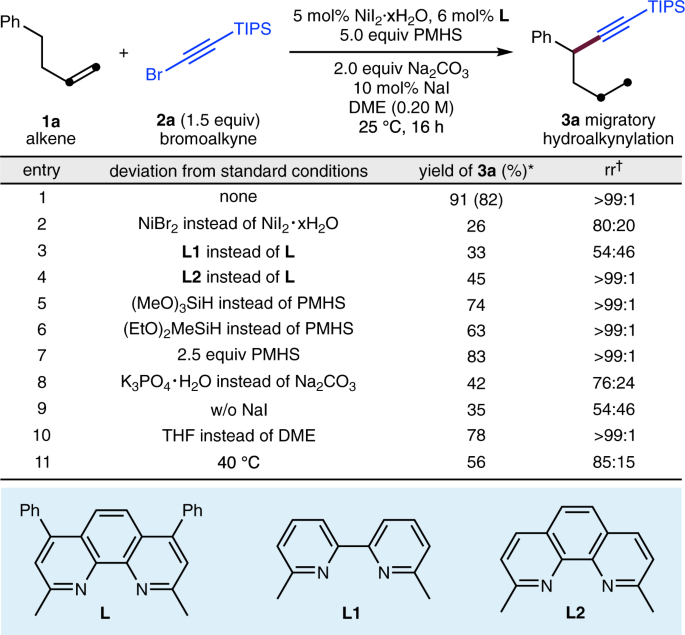
Nickel-catalysed migratory hydroalkynylation and enantioselective hydroalkynylation of olefins with bromoalkynes | Nature Communications

Regio- and enantioselective remote hydroarylation using a ligand-relay strategy | Nature Communications

Facile Synthesis of Chiral Arylamines, Alkylamines and Amides by Enantioselective NiH‐Catalyzed Hydroamination - Meng - 2021 - Angewandte Chemie - Wiley Online Library
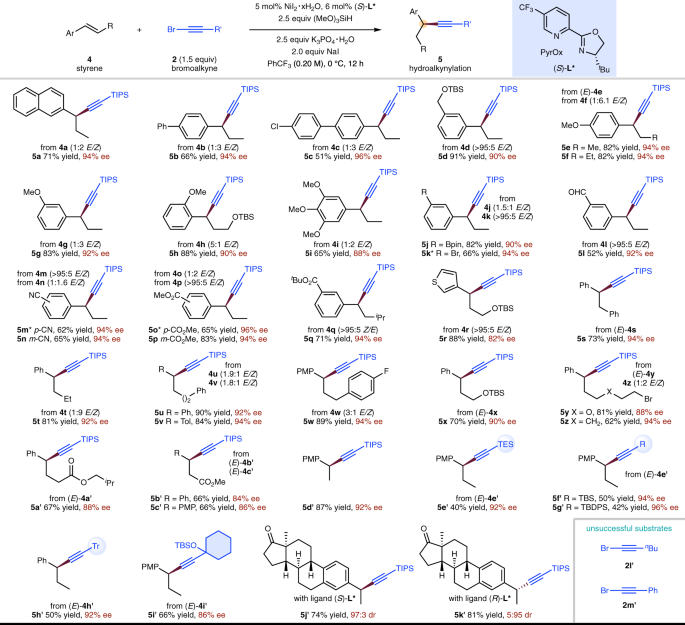
Nickel-catalysed migratory hydroalkynylation and enantioselective hydroalkynylation of olefins with bromoalkynes | Nature Communications
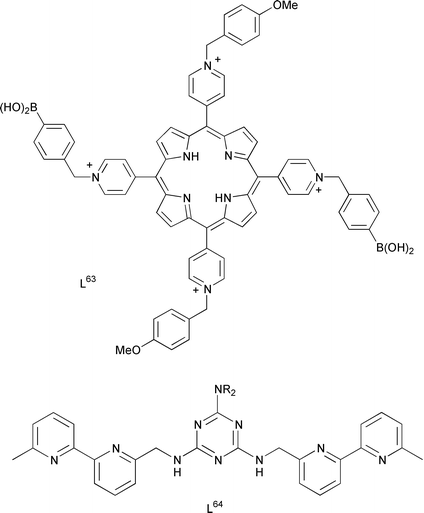
18 Supramolecular coordination chemistry - Annual Reports Section "A" (Inorganic Chemistry) (RSC Publishing) DOI:10.1039/B102906C

Nickel-Catalyzed Reductive Cross-Couplings: New Opportunities for Carbon–Carbon Bond Formations through Photochemistry and Electrochemistry | CCS Chemistry
18 Supramolecular coordination chemistry - Annual Reports Section "A" (Inorganic Chemistry) (RSC Publishing) DOI:10.1039/B102906C

Copper-Catalyzed Enantioselective Propargylic Amination of Propargylic Esters with Amines: Copper−Allenylidene Complexes as Key Intermediates | Journal of the American Chemical Society

Facile Synthesis of Chiral Arylamines, Alkylamines and Amides by Enantioselective NiH‐Catalyzed Hydroamination - Meng - 2021 - Angewandte Chemie - Wiley Online Library

Visible Light Photocatalysis: Applications and New Disconnections in the Synthesis of Pharmaceutical Agents | Organic Process Research & Development

C/Meo Collective Women's Crossover Front Knit Wrap Sweater Top, Grey Sculpt, XS at Amazon Women's Clothing store

Transition-Metal (Pd, Ni, Mn)-Catalyzed C–C Bond Constructions Involving Unactivated Alkyl Halides and Fundamental Synthetic Building Blocks | Accounts of Chemical Research

Nickel-catalysed migratory hydroalkynylation and enantioselective hydroalkynylation of olefins with bromoalkynes. - Abstract - Europe PMC

Covalent Modifiers: A Chemical Perspective on the Reactivity of α,β-Unsaturated Carbonyls with Thiols via Hetero-Michael Addition Reactions. - Abstract - Europe PMC

The steroid glycoside H.g.-12 from Hoodia gordonii activates the human bitter receptor TAS2R14 and induces CCK release from HuTu-80 cells | American Journal of Physiology-Gastrointestinal and Liver Physiology
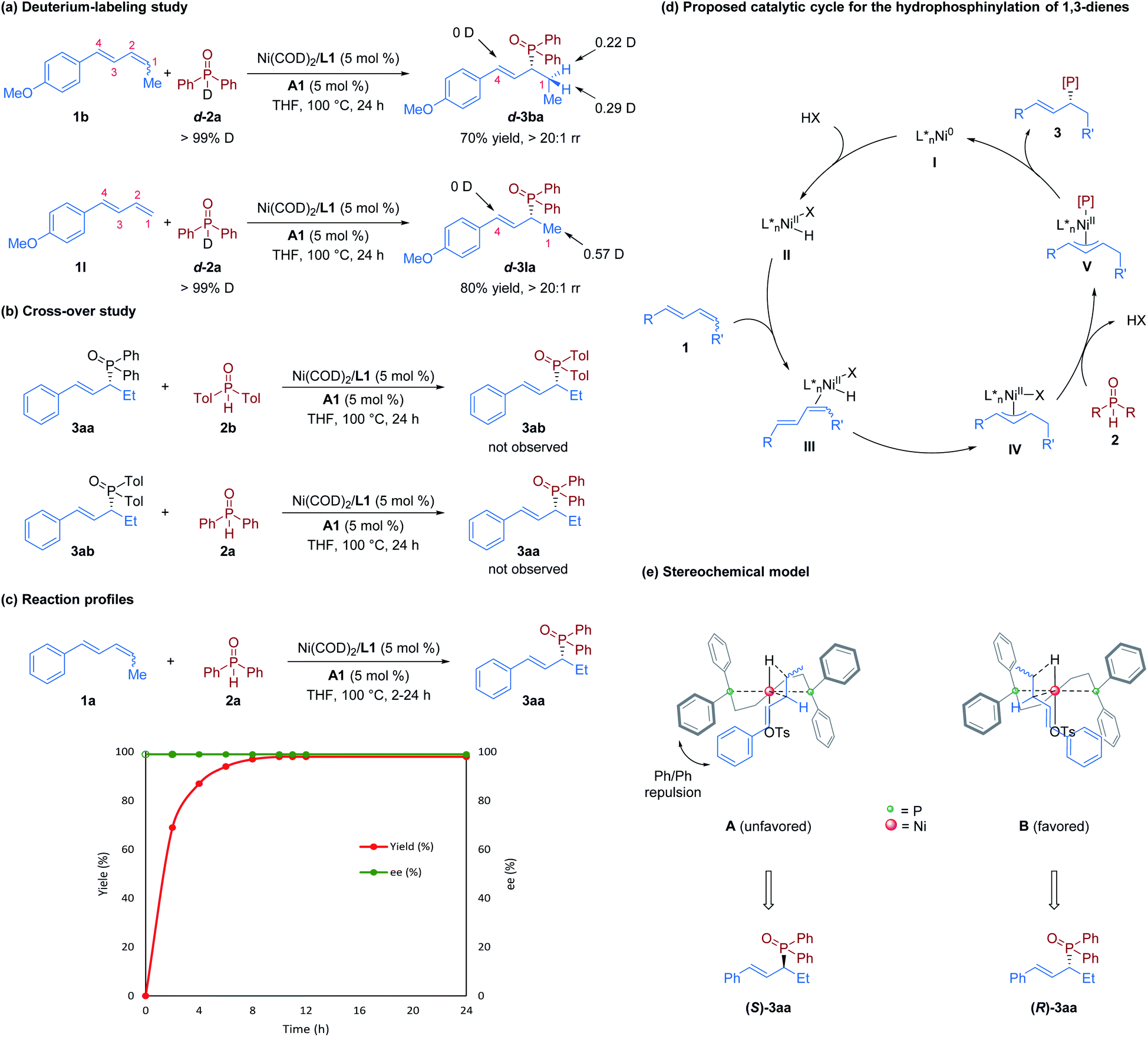
Nickel/Brønsted acid dual-catalyzed regio- and enantioselective hydrophosphinylation of 1,3-dienes: access to chiral allylic phosphine oxides - Chemical Science (RSC Publishing) DOI:10.1039/D1SC05651D

Low-Valent, High-Spin Chromium-Catalyzed Cleavage of Aromatic Carbon–Nitrogen Bonds at Room Temperature: A Combined Experimental and Theoretical Study | Journal of the American Chemical Society

A biomimetic SH2 cross-coupling mechanism for quaternary sp3-carbon formation. - Abstract - Europe PMC